
SEMI
Side Effects May Include
Evidence-based drug safety analysis. Compare what drug manufacturers report with what patients actually experience. Aggregates data from DailyMed, FAERS, VAERS, SIDER, and more.
Screenshots
Features
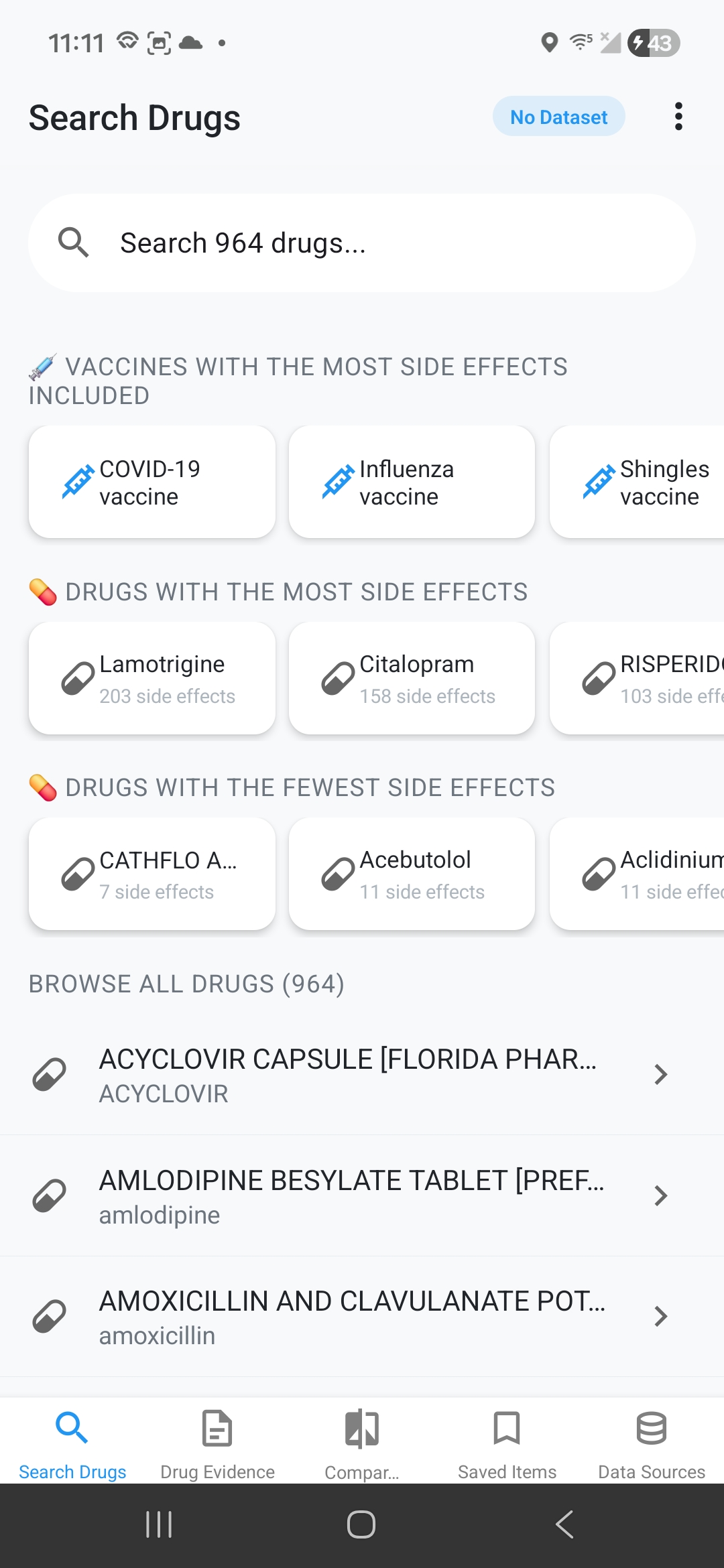
Intelligent Drug Search
Search thousands of medications with auto-complete, filter by name/generic/manufacturer, quick access to vaccines, browse drugs by side effect counts, and recent search history.
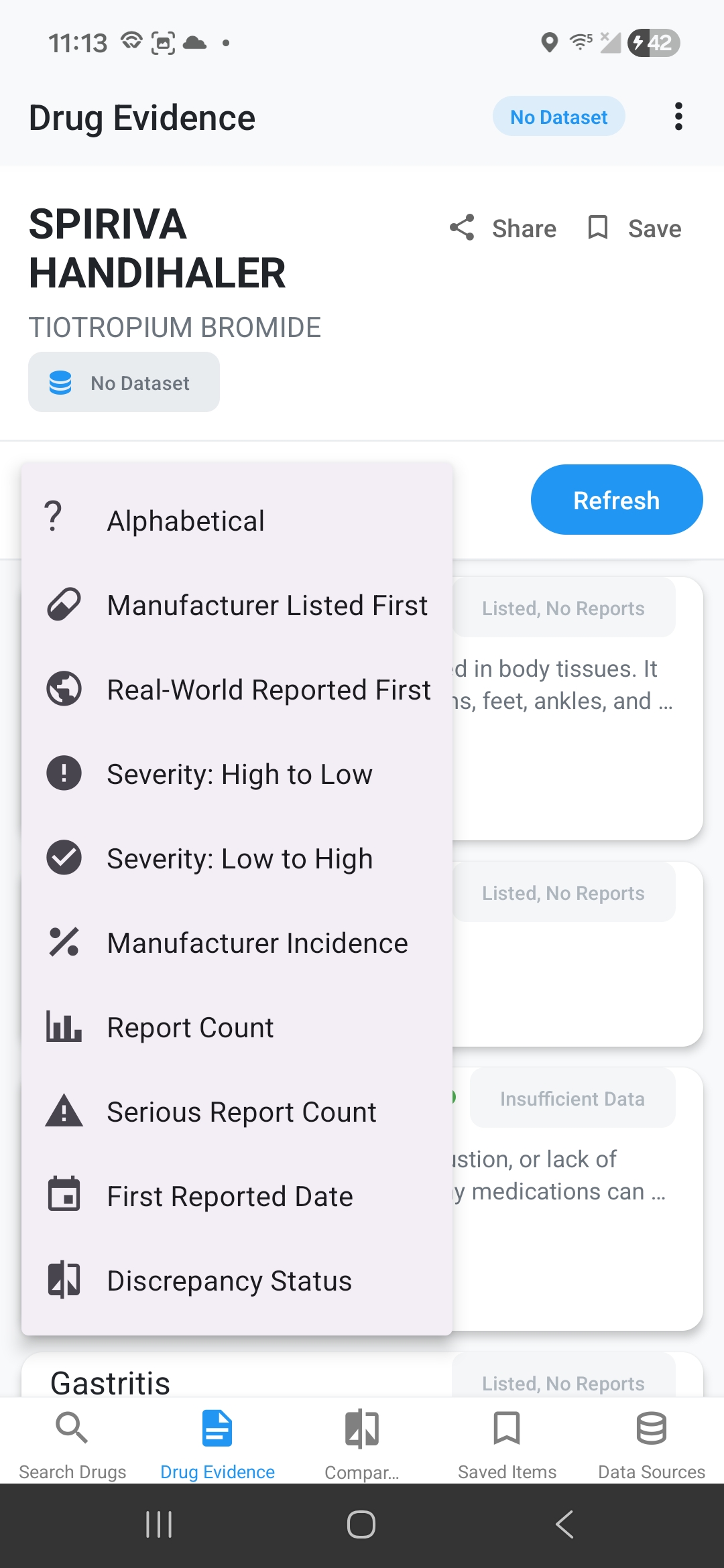
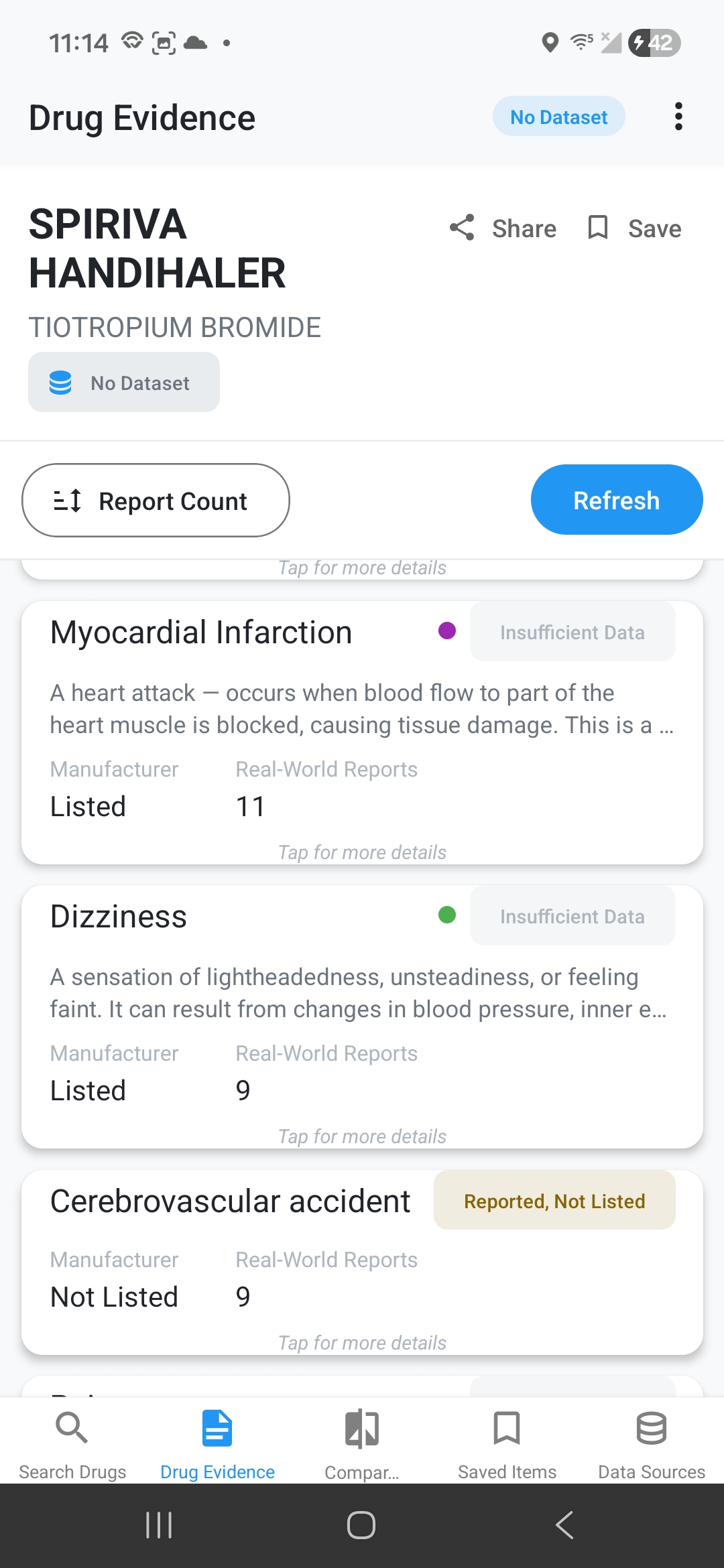
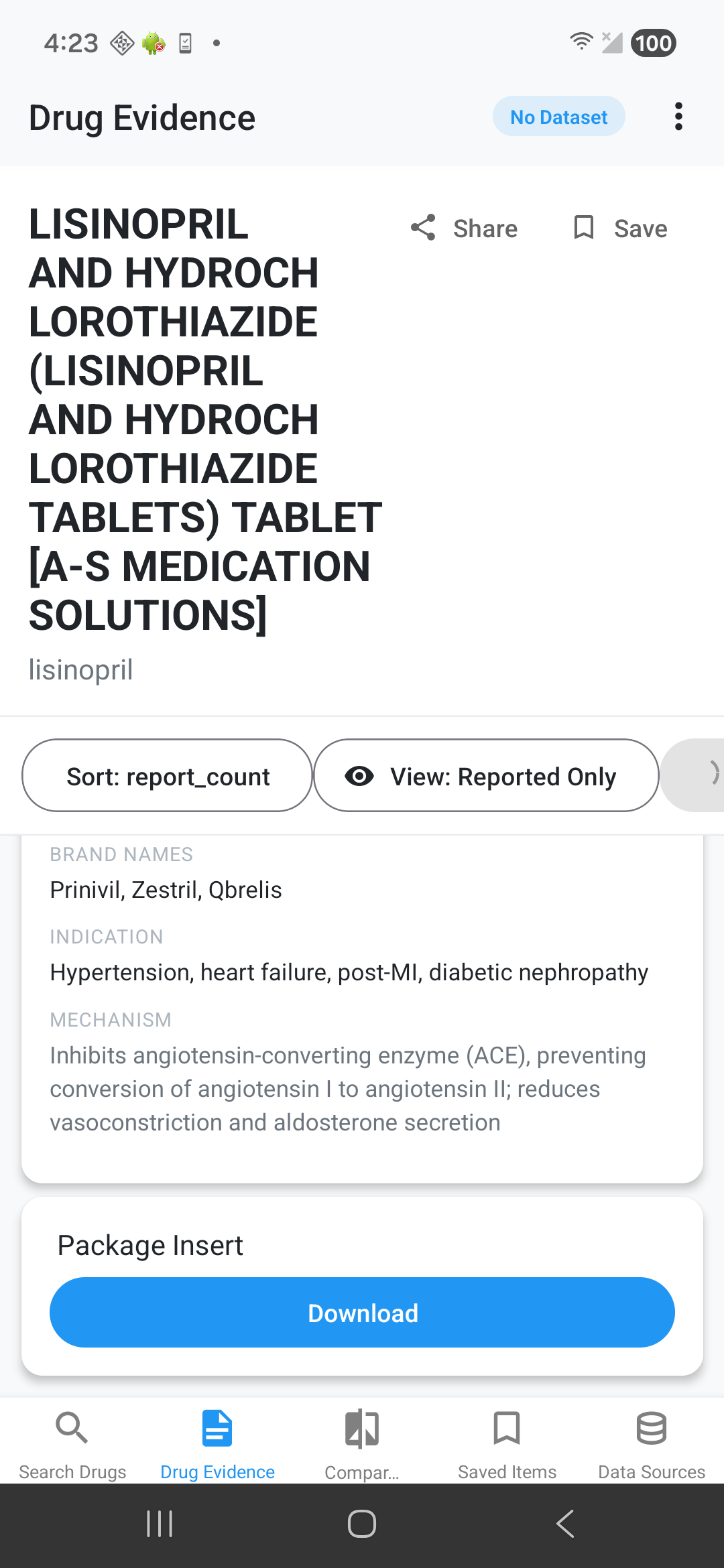
Side Effect Evidence Comparison
See discrepancy indicators: ✅ Match, ⚠️ Reported Not Listed, 📈 Higher Real-World Rate, ℹ️ Listed No Reports, 📉 Listed Lower Rate. Tap for detailed medical definitions.
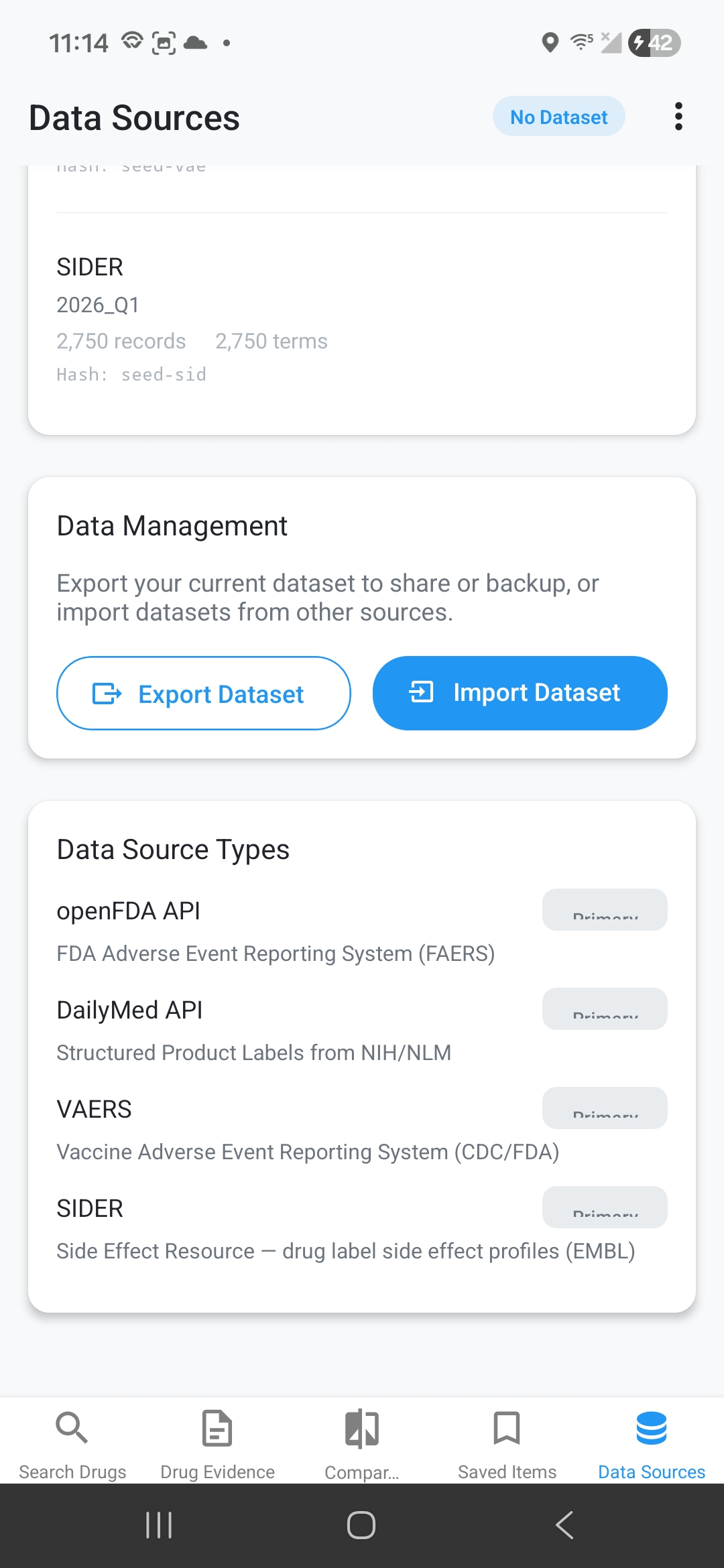
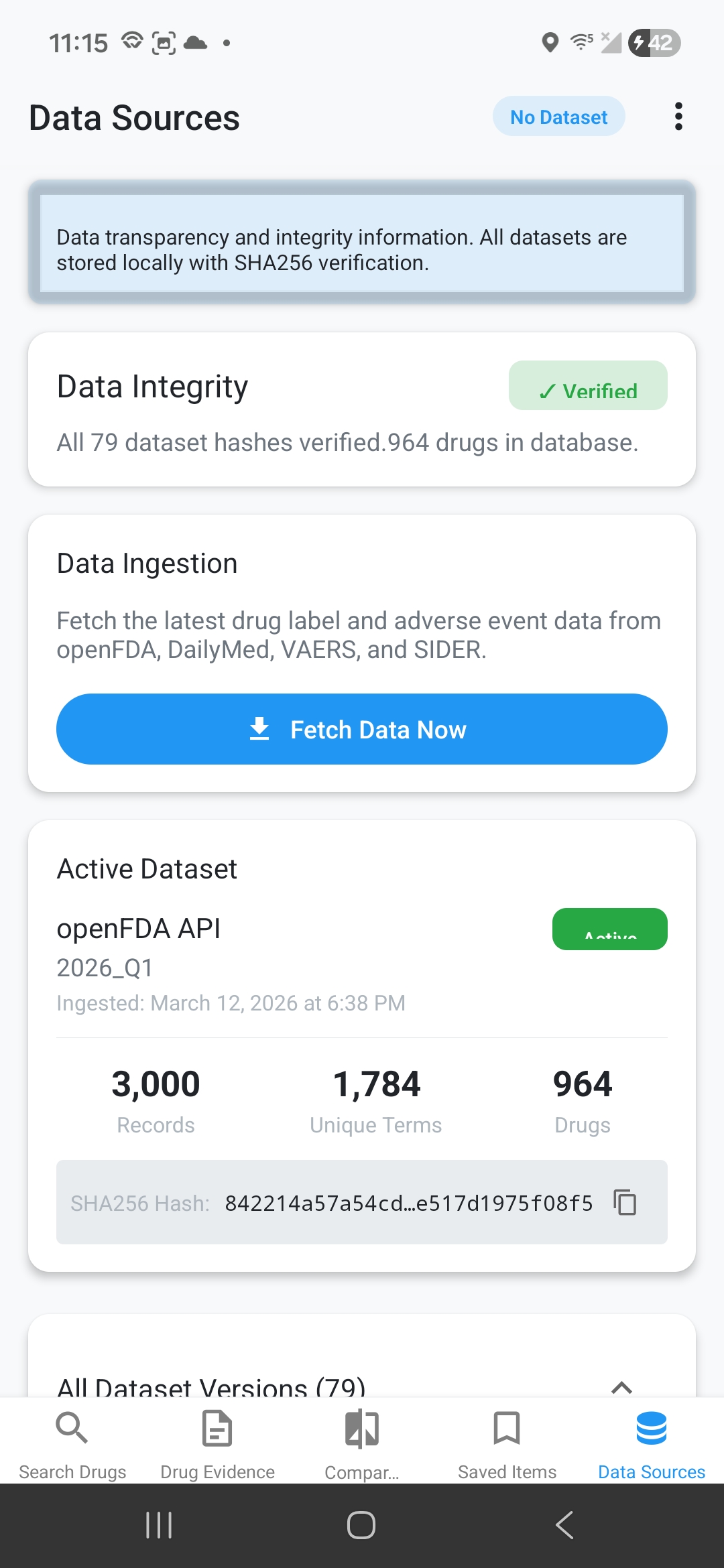
Data Source Transparency
Every piece of data has clear provenance: ingestion date, source adapter, record counts, SHA256 hash for integrity, and reliability ratings (Primary/Secondary/Tertiary).
Dataset Version Comparison
Track changes between ingestion runs. Compare side effect counts, see added/removed entries, identify manufacturer updates, validate hash integrity. Perfect for reproducible research.
Import & Export
Export dataset snapshots as JSON with full metadata. Import datasets from collaborators, archived snapshots, or verified external sources for data sharing and verification.
Offline-First Design
All data stored locally on your device. No cloud sync, no accounts, no telemetry. Works completely offline after initial setup. Complete user privacy guaranteed.
About SEMI
Side Effects May Include bridges the gap between clinical trial data and real-world adverse events. Clinical trials provide controlled data, but once a drug reaches the market, millions of patients take it under diverse conditions. The FDA's Adverse Event Reporting System (FAERS) collects real-world data, but patients and clinicians rarely compare this with manufacturer-reported side effects.
SEMI aggregates authoritative data sources (DailyMed, FAERS, VAERS, SIDER), standardizes terminology, computes discrepancies, preserves data integrity with cryptographic hashes, and enables reproducible research through import/export.
What We Do & Don't Do
We DO: Show everything unfiltered, attribute every piece of data to its source, verify data integrity with SHA256, and enable you to draw your own conclusions.
We DON'T: Calculate safety scores, make medical recommendations, filter out "rare" events, apply algorithmic weighting, or collect user data.
Who Uses SEMI?
- Researchers: Post-market surveillance studies, data provenance documentation, educational tools.
- Healthcare Professionals: Patient counseling, adverse event assessment, quality improvement.
- Patients & Advocates: Informed decision-making, advocacy & reporting, research participation.
Get SEMI Today
Evidence-based drug safety analysis. Compare FDA labels with real-world adverse events. Offline-first, completely private.
Coming soon to app stores. Android users can sideload APK now.
